Meet Vyleesi
Introducing the first and only FDA-approved as-needed treatment for premenopausal women with acquired, generalized hypoactive sexual desire disorder (HSDD).
Vyleesi was developed to help you get back to sexual desire on your terms.
Here's how:
Increases sexual desire
And reduces related distress.
Taken when you choose
For romance, fun, or just because.
Starts working in ~45 minutes
night for most, 8-10 hours.*
No alcohol restrictions
Say cheers to your connection.
Do not use Vyleesi if you have:
controlled (uncontrolled hypertension)
(cardiovascular) disease


How does Vyleesi work?
It’s long been known that the brain is responsible for sexual desire.
Years of research have identified the brain’s pathways that stimulate sexual desire. In women experiencing HSDD, an imbalance of neurotransmitters (chemical messengers) results in low sexual desire.
Vyleesi acts in the brain on certain types of neurotransmitters, called melanocortin receptors, to help improve sexual desire. The exact way that Vyleesi improves sexual desire isn’t fully known.
See the desired results


Here’s what they experienced
Vyleesi increased sexual desire and reduced related distress.
After taking Vyleesi, they experienced continued improvements in their HSDD symptoms.
The most common side effects are nausea, flushing, injection site reactions, headache, and vomiting.
Nearly 80% of women who completed the clinical study volunteered for an additional one-year extension.
Want to find out if Vyleesi is
right for you?
We’ll help you connect online with a healthcare professional experienced in helping women with HSDD.
The Prescribery platform helps pharmaceutical manufacturers (like the people who make Vyleesi) connect independent providers (who can prescribe Vyleesi if it is medically appropriate) directly with potential patients like you. Making it convenient for you to connect from home, on your time. Plus, it’s just $29 for the consultation!




Passionately ever after …
Hear from women
in our clinical studies
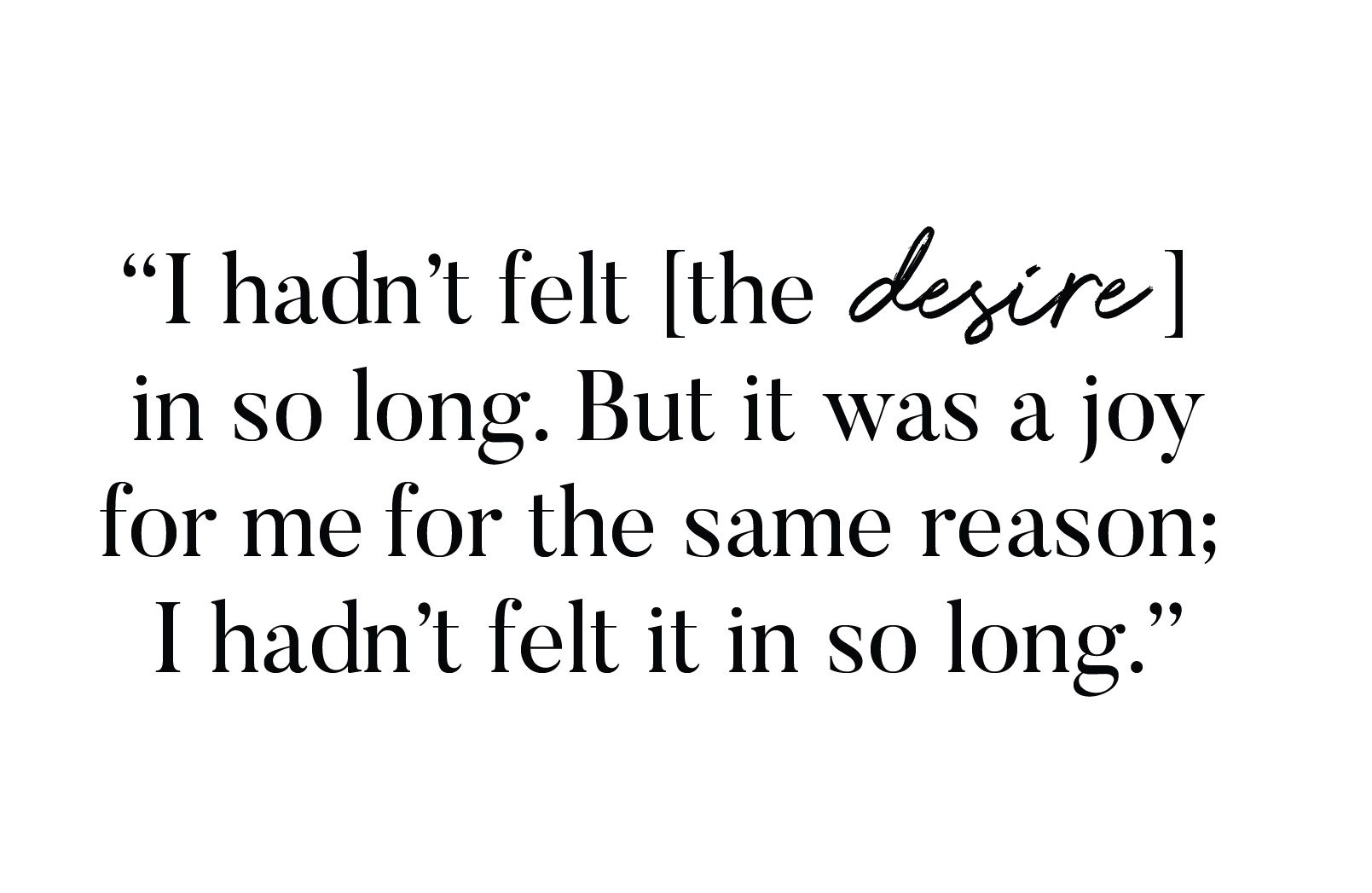
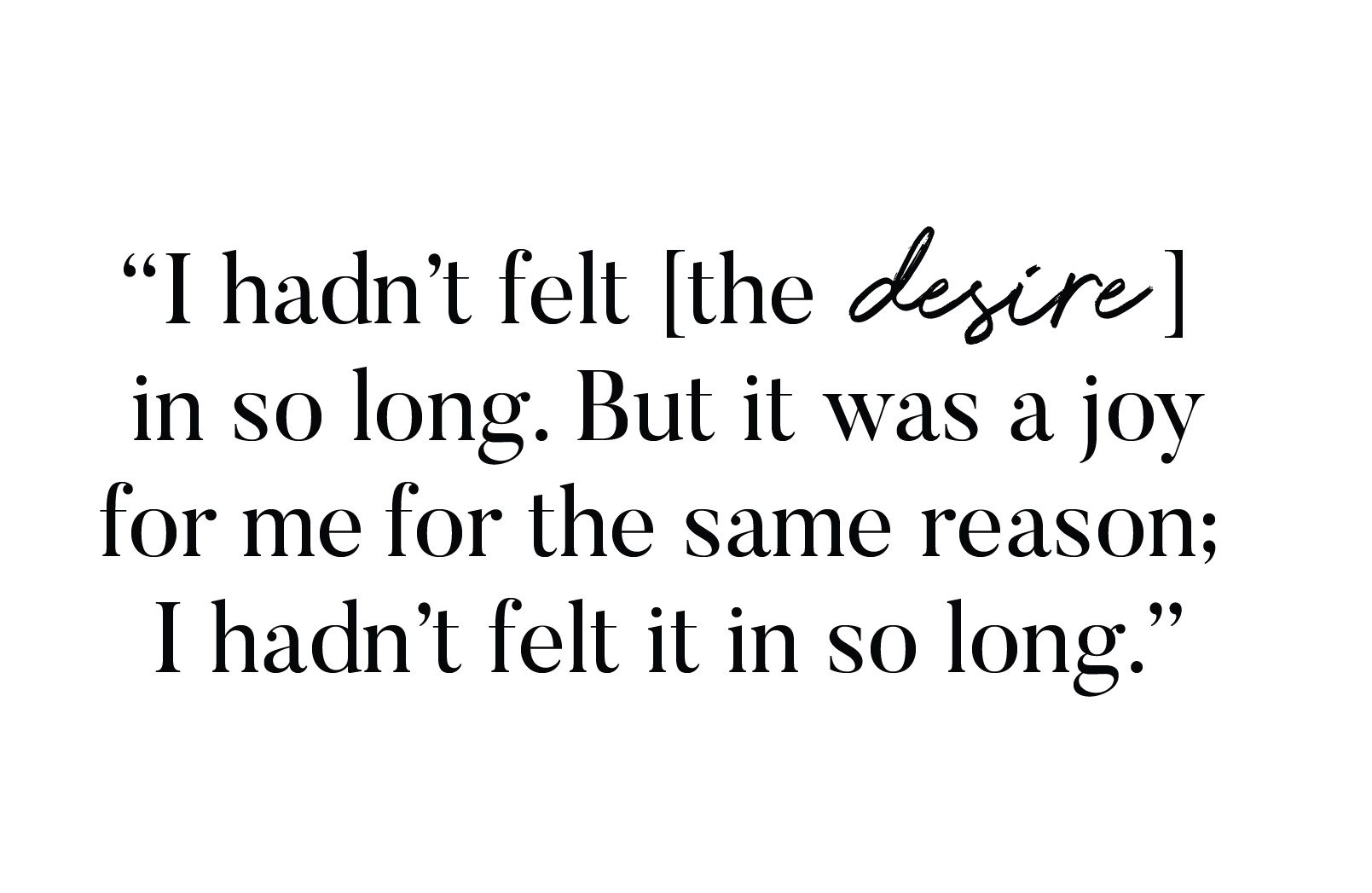




Real clinical trial participants


It can be hard to talk about sexual desire
We can help you start the conversation with your healthcare professional.
Footnotes
*Do not take Vyleesi more than one time within 24 hours or more than 8 times per month.
†Serious adverse reactions were reported in 1.1% of Vyleesi-treated patients and 0.5% of placebo-treated patients.
‡After completing the 24-week double-blind study period, 242 participants completed a voluntary exit survey to provide additional context to the clinical trial data. Consisting of 16 questions, the survey was conducted to understand the perceived effects and meaningfulness of Vyleesi. Additionally, a subset of the survey participants (80 women) also consented to be interviewed.

